Gujarat Board Textbook Solutions Class 10 Science Chapter 2 Acids, Bases and Salts
Gujarat Board Textbook Solutions Class 10 Science Chapter 2 Acids, Bases and Salts
GSEB Solutions Class 10 Science Chapter 2 Acids, Bases and Salts
Gujarat Board Class 10 Science Acids, Bases and Salts Textbook Questions and Answers
Question 1. A solution turns red litmus blue, its pH is likely to be –
(a) 1
(b) 4
(c) 5
(d) 10
Answer:
(d) 10
Question 2. A solution reacts with crushed egg-shells to give a gas that turns lime water milky. The solution contains:
(a) NaCl
(b) HCl
(c) LiCl
(d) KCl
Answer:
(b) HCl
Question 3. 10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount HCl solution (the same solution as before) required to neutralise it will be
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Answer:
(d) 16 mL
Question 4. Which one of the following types of medicines is used for treating indigestion?
(a) Antibiotic
(b) Analgesic
(c) Antacid
(d) Antiseptic
Answer:
(c) Antacid.
Question 5. Write word equations and then balance equations for the reaction taking place when:
(a) dilute sulphuric acid reacts with zinc granules.
(b) dilute hydrochloric acid reacts with magnesium ribbon.
(c) dilute sulphuric acid reacts with aluminium powder.
(d) dilute hydrochloric acid reacts with iron filings.
Answer:
(a) Zinc granule + dil. Hydrochloric acid → Zinc chloride + Hydrogen gas
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
(b) Magnesium+dil. Hydrochloric acid → Magnesium chloride + Hydrogen gas
Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)
(c) Aluminium + dil. Sulphuric acid → Aluminium sulphate + Hydrogen gas
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)
(d) Iron + dil. Hydrochloric acid → Iron chloride + Hydrogen
2Fe(s) + 6HCl(aq) → 2FeCl3(aq) + 3H2(g)
Question 6. Compounds such as alcohols and glucose also contain hydrogen but are not categorised as acids. Describe an activity to prove it.
Answer:
1. Take a cork with two nails fixed on it.
2. Keep this cork inside the beaker.
3. Connect the nails to the battery, bulb and key as shown in the figure.
4. Now add ethanol in it and record your observation repeat the same set up for glucose and record your observations.
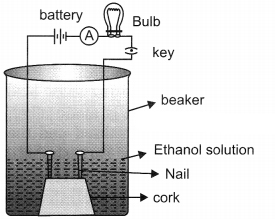
Observation: The bulb will not glow because charge is not flowing through it.
Conclusion: The experiment shows that glucose and ethanol do not ionise, (H are not released) therefore, they are not categorised as acids.
Question 7. Why does distilled water not conduct electricity, whereas rain water does?
Answer:
Distilled water is pure water and it does not form ions. Whereas rain water contains impurities in it like acid which contains ions and release them when dissolved in water. Hence, there are no ions in distilled water, so electricity is not conducted but there are ions in rain water so electricity is conducted.
Question 8. Why do acids not show acidic behaviour in the absence of water?
Answer:
Acids cannot release H+ ions in absence of water, only on dissolving in water acids release H+ ions. Acids show acidic behaviour only due to H+ ions released in presence of water.
Question 9. Five solutions A, B, C, D and E when tested with universal indicator showed pH as 4, 1, 11, 7 and 9 respectively. Which solution is
(a) neutral?
(b) strongly alkaline?
(c) strongly acidic?
(d) weakly acidic?
(e) weakly alkaline?
Arrange the pH in increasing order of hydrogen ion concentration.
Answer:
(i)
(a) ‘D’ with pH = 7 is neutral is strongly
(b) ‘C’ with pH = 11 alkaline is strongly
(c) ‘B’ with pH = 1 acidic is weakly
(d) ‘A’ with pH = 4 acidic is weakly
(e) ‘E’ with pH = 9 alkaline
(ii) pH in increasing order of hydrogen ion concentration
11 < 9 < 7 < 4 < 1
C < E < D < A < B
Question 10. Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. Amount and concentration taken for both the acids are same. In which test tube will the fizzing occur more vigorously and why?
Answer:
In test tube A hydrochloric acid is present which is a strong acid as compared to acetic acid present in test tube B. The fizzing occurs more vigorously in test tube A as HCl is strong and dissociates completely into H+ and Cl ions for the reaction.
Question 11. Fresh milk has pH of 6. How do you think the pH will change as it turns into curd? Explain your answer.
Answer:
pH of milk will decrease when it turns into curd due to the formation of lactic acid.
Question 12. A milkman adds a very small amount of baking soda to fresh milk.
- Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
- Why does this milk take a long time to set as curd?
Answer:
- pH is shifted to alkaline so that milk does not spoil by becoming sour by releasing lactic acid.
- Milk is made alkaline by adding baking soda so the lactic acid formed will get neutralised and therefore will take a little more time to set become acidic.
Question 13. Plaster of Paris should be stored in a moisture-proof container. Explain why?
Answer:
Plaster of Paris reacts with moisture/water to form gypsum.
Question 14. What is a neutralisation reaction? Give two examples.
Answer:
The reaction in which acid reacts with base (loses their property) to form salt and water is called neutralisation reaction.
Example:
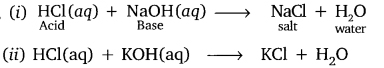
Question 15. Give two important uses of washing soda and baking soda.
Answer:
Washing soda:
- It is used in glass, soap and paper industries.
- It is used to remove permanent hardness of water.
Baking Soda:
- It is used as an antacid.
- It is used in soda-acid fire extinguishers.
Gujarat Board Class 10 Science Acids, Bases and Salts Additional Important Questions and Answers
Short Answer Type Questions
Question 1. What happens when
(a) Baking soda is heated
(b) Blue coloured copper sulphate crystals are heated
(c) Water is added to lime?
Answer:
(a) When baking soda is heated it forms sodium carbonate, water and releases carbon dioxide gas
![]()
(b) Blue coloured copper sulphate on heating becomes colourless as it loses water of crystallisation.
![]()
(c) On adding water to lime heat is evolved and slaked lime is formed.
CaO + H2O → Ca(OH)2+ heat
Question 2. A white powder which sets hard on adding water is also used in hospitals. Name this powder. How is it prepared? Write the chemical reaction involved in its preparation.
Answer:
The white powder is Plaster of Paris (CaSO4.1/2 H2O)

It is prepared by heating gypsum at 393 K.
Question 3. Write balanced chemical equations for the following:
- Calcium carbonate reacts with hydrochloric acid
- Dilute sulphuric acid reacts with zinc granules
- Calcium oxychloride reacts with hydrochloric acid.
Answer:
- CaCO3 + 2HCl → CaCl2 + H2O + CO2↑
- dil. H2SO4 + Zn → ZnSO4 + H2
- CaOCl2 + 2HCl → CaCl2 + H2O + Cl2
Question 4. Name the ions present in the following salts. Name the acid and base from which they can be obtained. magnesium sulphate, sodium carbonate, potassium chloride.
Answer:
The ions present in the compounds are:
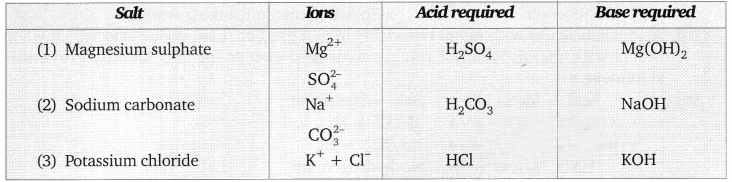
Question 5. Give three ways in which salts can be prepared.
Answer:
Salts can be obtained in the following ways:
(i) Acid reacts with base to give salt and water.
![]()
(ii) Metals react with acids to form salt and give hydrogen gas.
Mg + 2HCl → MgCl2 + H2
(iii) Metallic oxide reacts with acid to form salt.
CuO + 2HCl → CuCl2 + H2O
Question 6. Give one example for each of the following acids salts – chloride salts, carbonate salts and sulphate salts.
Answer:
- Chloride salts → Magnesium chloride, Calcium chloride
- Carbonate salts → Sodium carbonate, Potassium carbonate
- Sulphate salts → Calcium sulphate, Magnesium sulphate
Question 7. Name the acid present in the following:
Vinegar, Lemon, Tomato, Tamarind, Orange, Curd.
Answer:
Source:
- Vinegar
- Lemon
- Tomato
- Tamarind
- Orange
- Curd
Acid:
- Acetic acid
- Citric acid
- Oxalic acid
- Tartaric acid
- Citric acid
- Lactic acid
Question 8. Name the properties responsible for the following uses of baking powder:
- Baking industry
- As an antacid
- As soda-acid fire extinguisher
Answer:
Properties of baking powder:
- On heating releases CO2 gas
- Alkaline in nature, neutralises excess acid in stomach
- When it reacts with acid it releases CO2 gas which can extinguish fire.
Uses:
- Baking industry Antacid
- Soda-acid
- fire extinguisher
Question 9. Give the properties and uses of bleaching powder.
Answer:
Properties-Oxidising agent Uses:
- Bleaching – Used in bleaching cotton, wood pulp, clothes.
- Oxidising agent – Used in chemical industries.
- Disinfectant – To kill germs in drinking water.
Question 10. Acid when reacts with metal release hydrogen gas but there is one acid which when reacts with metal does not release hydrogen except for two metals. Prove this statement.
Answer:
Acid + Metal → salt + hydrogen
2HCl + 2Na → 2Nacl + H2
H2SO4 + 2Na → Na2SO4 + H2
HNO3 + Na → No hydrogen
Nitric acid does not release hydrogen gas when it reacted with metals. This is because nitric acid is strong oxidising agent. Nitric acid reacts only with magnesium and manganese to evolve hydrogen gas
Mg + 2HNO3 → Mg(NO3)2 + H2 ↑
Mn + 2HNO3 → Mn(NO3)2 + H2 ↑
Question 11. Give six uses of acids.
Answer:
Uses of acids are:
- In storage batteries (H2SO4)
- As food preservative (acetic acid)
- In making baking powder (tartaric acid)
- In manufacturing of fertilizers (nitric acid)
- In making PVC (Poly vinyl chloride) (Hydrochloric acid)
- As bathroom cleaner (hydrochloric acid)
Question 12. Give six uses of bases.
Answer:
Uses of bases are:
- In making soap (NaOH)
- As an antacid (Mg(OH)2)2
- In making bleaching powder (Ca(OH)2)
- In removing acidity of soils (Ca(OH)2)
- In white washing (Ca(OH)2)
- In making fertilizers (NH4OH)
Question 13. Give six uses of salts.
Answer:
Uses of salts are:
- Adds taste to food (NaCl)
- In making a feerzing mixture (Nacl)
- As washing soda for clothes (Na2CO3)
- As baking soda (NaHCO3)
- To purify water (alum, CaOCl2)
- In making of soaps (Nacl)
Question 14. Four samples A, B, C and D were given to test their nature. A student found the change in pH paper as follows:
A → green colour C → blue colour
B → orange colour D → pink colour
Find the nature of given sample.
Answer:

Long Answer Type Questions
Question 1. What are acids? Give three properties of acids. Give the test for acids.
Answer:
Acids are those substances which release hydrogen ions (H+) when dissolved in water.
Properties of Acids:
- Acids are sour in taste.
- Acids conduct electricity when dissolved in water.
- Acids react with metals to from salt and hydrogen gas.
Test for Acid:
- Turns blue litmus red
- Methyl orange turns red in acid
Question 2. Design an activity to prove that acids show acidic behaviour only when dissolved in water.
Answer:
Take a clean test tube and add solid NaCl in it. Add some concentrated sulphuric acid in it. Fix the mouth of the test tube with cork and fix delivery tube in it. The reaction begins and gas comes out from the delivery tube, it is HCl gas. Take a litmus paper (dry) and test the presence of gas, there will be no colour change
Now wet the litmus paper and bring it near the mouth of delivery tube. The blue litmus paper turns red. This activity proves that hydrogen ions in HCl are produced in the presence of water.
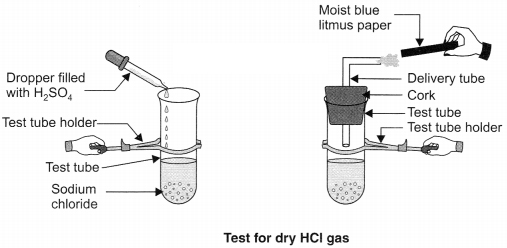
Question 3. How do acids and bases react with each other? Explain with an activity.
Answer:
Take a test tube with 2 mL of base in it i.e., -NaOH solution, add 2 drops of phenolphthalein solution as an indicator. The solution turns pink.
Now add HCl solution to this test tube drop by drop, the pink colour starts disappearing. At this point we say that the acid reacts with base to become neutral. On adding few drops of NaOH to the neutral solution obtained above, the pink colour of phenolphthalein reappear. Again add few drops of HCl, the colour disappears. This shows that acids react with bases to form (neutral) salt and water.
Question 4. What is bleaching powder? How is bleaching powder produced? Give its chemical . equation and write its three uses.
Answer:
Bleaching powder is calcium oxychloride, CaOCl2.
Preparation: On passing chlorine gas through a test tube containing dry slaked lime Ca(OH)2, bleaching powder is produced.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
Uses:
- It is used as bleaching agent in textile industry and paper factories.
- It is used as an oxidising agent in chemical industries.
- It is used as disinfectant for drinking water.
Question 5. What is baking soda and baking powder chemically? What would happen if we add baking soda in making cakes instead of baking powder?
Answer:
Baking soda is NaHCO3 – Sodium hydrogen carbonate
Baking powder is NaHCO3 + Tartaric acid.
If we add baking soda in making of cakes on heating it will produces sodium carbonate which will add bitter taste to the cake. To avoid this, tartaric acid is added to baking soda which produces sodium salt of acid and does not change the taste of cake.
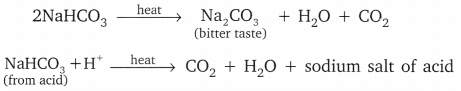
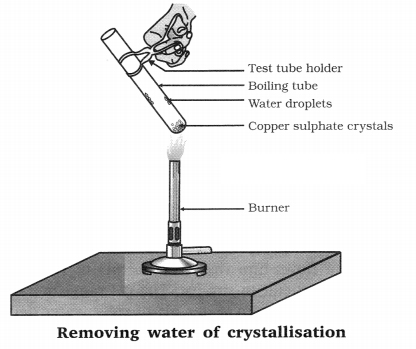
Question 6. Crystals of salt are formed by holding some water molecules. How can you show that crystals of salt are not really dry?
Answer:
To show that crystals of salt are not dry:
Take a dry test tube, add few crystals of copper sulphate (blue colour). Fix the test tube in a stand and heat it on flame. The blue colour of copper sulphate becomes colourless and drops of water are seen on the inner sides of test tube.
This shows that crystals are not dry they hold water molecules.
Practical Based Questions (Solved)
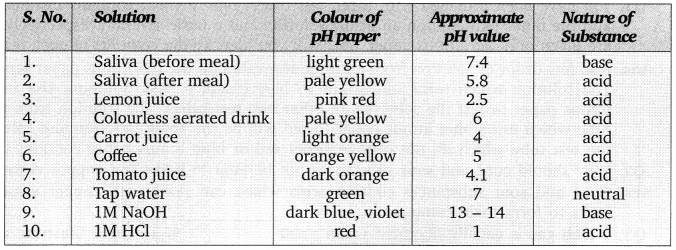
Question 1. A student wants to test the pH of human saliva at different times. Suggest the right time to collect the pH of it for wide variety and why?
Answer:
The student should collect the pH of saliva once in the morning without brushing the teeth, then after brushing teeth, before any meal and after meals. The pH of mouth changes when we eat food and also when we brush our teeth.
Question 2. In the lab a test tube rack is placed with test tubes containing some acids in it. How will you classify these acids?
Answer:
To classify the acids we should test their pH and group them as strong or weak acids.
Question 3. What is the pH of water? Does it change on heating the water? Explain.
Answer:
The pH of pure water is generally 7 at 25 degree Celcius. On heating water the pH will change, it decreases because on heating water molecules will ionise more and thus concentration of H+ (and OH–) increases.
Question 4. A test was conducted in the lab to find the pH of different cold drinks. Draw the observation table to collect the data for this experiment and predict the result for the same.
Answer:
Observation table to collect data:

Prediction: The pH of the dark cold drink will be higher as it contains more caffeine which is more acidic and the cold drink which releases more bubbles on opening is highly acidic and will have high pH as the carbonated water is present in it.
Question 5. A student wants to test the pH of few salts present at home like baking soda, common salt, washing powder, bleaching powder and eon salt. How can he find the pH of dry salts? Explain
Answer:
The pH of dry salts cannot be obtained hence all these salts need to be dissolved in distilled water to get their pH and the samples can be tested using pH paper.
Question 6. Four juices were taken in different test tubes to find their pH, the colour changes on the pH paper were recorded as pink, orange, yellow and green. What conclusion can you draw from this data.
Answer:
The sample that gave pink color has lower pH i.e. more acidic then the others and the sample that gave orange and yellow colour has higher pH i.e. it is less acidic than the others. The sample with green colour change is neutral.
Question 7. In one of the projects at school students were told to make natural indicators to test the samples for their nature. Suggest two natural indicators students can make easily.
Answer:
The students can make the natural indicators using red cabbage or hibiscus flower. On crushing the red cabbage or hibiscus and making the juice it can be used as natural indicator as they change colour and give different result in acid and bases.
Gujarat Board Class 10 Science Acids, Bases and Salts InText Questions and Answers
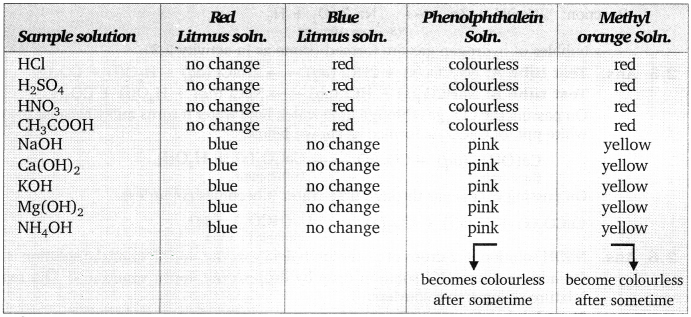
Question 1. You have been provided with three test tubes, one of them contains distilled water and the other two contain an acidic solution and a basic solution respectively. If you are given only red litmus paper, how will you identify the contents of each test tube?
Answer:
Add few drops of solution from all three test tubes on the red litmus paper separately. The solution which turns red litmus to blue contains basic solution. Use this blue litmus paper to test the solutions in other two test tubes. The solution from the test tube which turns blue litmus paper to red will be the acidic solution and solution of the test tube which do not change either red or blue litmus paper contains water.
Question 2. Why should curd and sour substances not be kept in brass and copper vessels?
Answer:
Curd and sour substances contain acids which can react with copper vessels and brass to form toxic compounds.
Question 3. Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Answer:
When an acid react with a metal it liberates hydrogen gas.
Example:
Zn(s) + 2HCl(aq) → ZnCl2(aq)+ H2(g)
To test the presence of H2 gas, bring a burning match stick near the mouth of the test tube where H2 gas is released, the match stick burns with a pop sound.
Question 4. Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Answer:
Metal compound A is CaCO3
Gas evolved is CO2
Balanced equation: CaCO3(s) + 2HCl(aq) → CaCl2(aq) + CO2(g) + H2O(l)
Question 5. Why do HCl, HNO3 etc., show acidic characters in aqueous solutions while solutions; of compounds like alcohol and glucose do not show acidic character?
Answer:
Solutions like HCl, HNO3 etc. get ionised in aqueous solutions and due to the presence of H+ ions they show acidic characters. While solutions of compounds like alcohol and glucose do not form any such ions so they do not show acidic characters.
Question 6. Why does an aqueous solution of an acid conduct electricity?
Answer:
Acid when forms a solution in water gets ionised, due to the presence of these ions electricity is conducted through it.
Question 7. Why does dry HCl gas not change the colour of the dry litmus paper?
Answer:
Dry HCl gas does not release H+ ions and hence, the acidic property of gas is not imparted.
Question 8. While diluting an acid, why is it recommended that the acid should be added to water and not water to the acid?
Answer:
When acid and water mix, the reaction is highly exothermic, the acid may splash, cause burns and even the bottle/container can break. To avoid this and allow the heat evolved to be absorbed by water slowly, acid is added to water for diluting it.
Question 9. How is the concentration of hydronium ions (H3O+) affected when a solution of an acid is diluted?
Answer:
When the solution-of acid is diluted then the concentration of hydronium ion (H3O+) per unit volume decreases.
Question 10. How is the concentration of hydroxide ions (OH–) affected when excess base is dissolved in a solution of sodium hydroxide?
Answer:
When base is mixed with water there is decrease in the concentration of OH– ions per unit volume.
Question 11. You have two solutions A and B. The pH of solution A is 6 and pH of solution B is 8. Which solution has more hydrogen ion concentration? Which of this is acidic and which one is basic?
Answer:
A with pH = 6 is acidic B with pH = 8 is basic
‘A’ has more hydrogen ion concentration.
Question 12. What effect does the concentration of H+ (aq) ions, have on the nature of the solution?
Answer:
If a solution has higher concentration of H+ ions it is more acidic in nature.
Question 13. Do basic solutions also have H+ (aq) ions? If yes, then why are these basic?
Answer:
Yes basic solutions also have H+ ions, but they are basic in nature due to more number of OH– ions.
Question 14. Under what soil condition do you think a farmer would treat the soil of his fields with quick lime (calcium oxide) or slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Answer:
When the soil is acidic in nature, the farmer would add quicklime (CaO) or slaked lime (Ca(OH)2) or chalk (CaCO3)to make it neutral.
Question 15. What is the common name of the compound CaOCl2?
Answer:
The common name of CaOCl2 is bleaching powder and chemical name is calcium oxychloride.
Question 16. Name the substance which on treatment with chlorine yields bleaching powder.
Answer:
Calcium hydroxide (Ca(OH)2) when treated with chlorine yields bleaching powder.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
Question 17. Name the sodium compound which is used for softening hard water.
Answer:
Sodium carbonate.
Question 18. What will happen if a solution of sodium hydrocarbonate is heated? Give the equation of the reaction involved.
Answer:
When sodium hydrocarbonate is heated, sodium carbonate, water, and carbon dioxide gas is obtained.
![]()
Question 19. Write an equation to show the reaction between Plaster of Paris and water.
Answer:
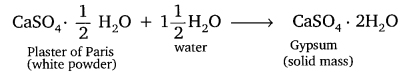
In-Text Activities Solved
Activity 2.1
Answer:
Activity 2.2
Answer:
Cloth strips when kept in a bag with finely chopped onions and left overnight in the fridge, the strips would smell of onion.
Cloth strip + dilute HCl Solution → We can smell onion, red colour of cloth strip changes to pale red.
Cloth strip + dilute NaOH solution → Red colour of cloth strip changes to green.
Odour test:
On adding vanilla essence → In dilute NaOH → no smell In dilute HCl → Smell of vanilla essence exists
On adding clove oil → In dilute HCl → smell of clove exists
In dilute NaOH → smell of clove does not exist
Question 2.3
Answer:
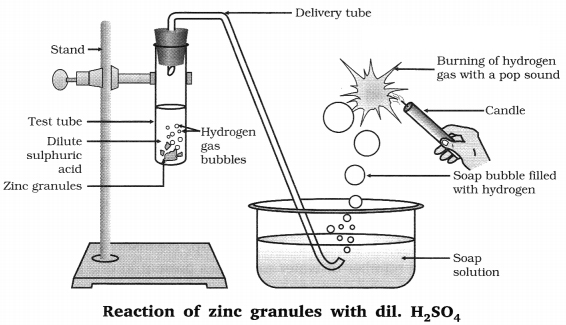
5 mL of dilute sulphuric acid and zinc granule → on surface of zinc granules-bubbles of hydrogen gas are formed.
On passing the hydrogen gas evolved through a soap solution → bubbles are formed due to low surface tension of soap solution.
On taking a burning candle near a gas filled bubbles → gas burns with a pop sound.
All other acids HCl, HNO3 and CH3COOH shows the same observation.
Activity 2.4
Answer:
2 mL of NaOH solution + granulated zinc.
![]()
→ bubbles of hydrogen gas are formed (same as in activity 2.3).
Activity 2.5
Answer:
Test tube A: Na2CO3(s) + 2HCl (aq) → 2NaCl (aq) + H2O(Z) + CO2(g)
Test tube B: NaHCO3(s) + HCl (aq) → NaCl (aq) + H2O(l) + CO2(g)
On passing the CO2 gas through lime water, lime water it’turns milky because insoluble white ppt. of CaCO3 is formed as shown below.
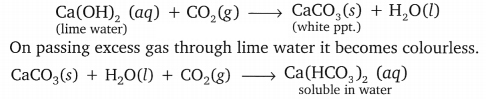
Activity 2.6
Answer:
NaOH solution + 2 drops of phenolphthalein solution → Colour of the solution is pink.
On adding dilute HCl solution drop by drop to the above solution → The reaction mixture changes to colourless.
Phenolphthalein colour changes from pink to colourless as neutralisation of HCl and NaOH takes place.
On adding few drops of NaOH to the above mixture, pink colour reappears because initially the effect of base was nullified by an acid and so the colour changed to colourless but on addition of some more base the pink colour of phenolphthalein reappeared.
Activity 2.7
Answer:
When dilute hydrochloric acid is added to copper oxide solution, the colour of the solution becomes blue-green and the copper oxide dissolves. The blue-green colour is due to the formation of copper (II) chloride.
![]()
Activity 2.8
Answer:
In the given set up, when different solutions are poured in the beaker separately, following observations were recorded:

Acid solution in water conducts electricity

Activity 2.9
Answer:
On adding concentrated sulphuric acid to the test tube containing NaCl, the reaction takes place and dry HCl gas is produced.
On testing this gas with litmus paper following observations were recorded:
HCl gas → Dry litmus paper → No change of colour of blue litmus paper
HCl gas → Wet litmus paper → blue litmus paper changes to red
This activity infers that only HCl solution releases H+ ions and the acidic property exists due to H+ ions.
Activity 2.10
Answer:
On adding a few drops of concentrated H2SO4 to the water in a beaker, it becomes hot as the reaction is highly exothermic.
On adding NaOH pellets to water → the beaker becomes hot, reaction is exothermic.
Activity 2.11
Answer:
Activity 2.12
Answer:
To test the pH of soil, take 2g soil in a test tube, add 5 mL water and shake it. Filter the contents, collect the filtrate to check the pH.
If pH of soil is 7: It is neutral – plants grow well
If pH is less than 7: It is acidic – plants are affected
If pH is more than 7: It is basic – plants are affected
Activity 2.13
Answer:
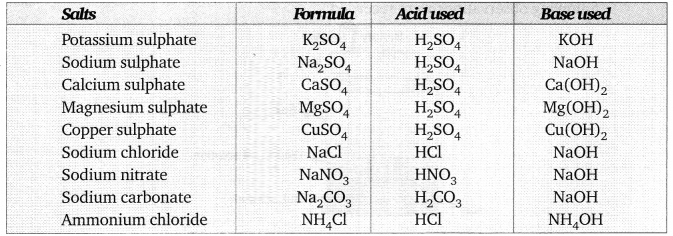
Formulae of the salts:
Activity 2.14
Answer:
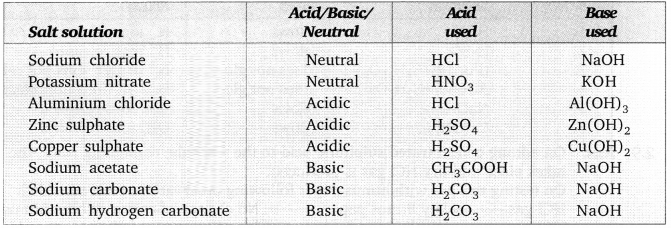
Activity 2.15
Answer:
On heating blue crystals of copper sulphate, it becomes colourless and few drops of water are seen on the test tube.
On adding few drops of water to the heated (anhydrous) copper sulphate, the blue colour of copper sulphate reappears.
Follow on Facebook page – Click Here
Google News join in – Click Here
Read More Asia News – Click Here
Read More Sports News – Click Here
Read More Crypto News – Click Here